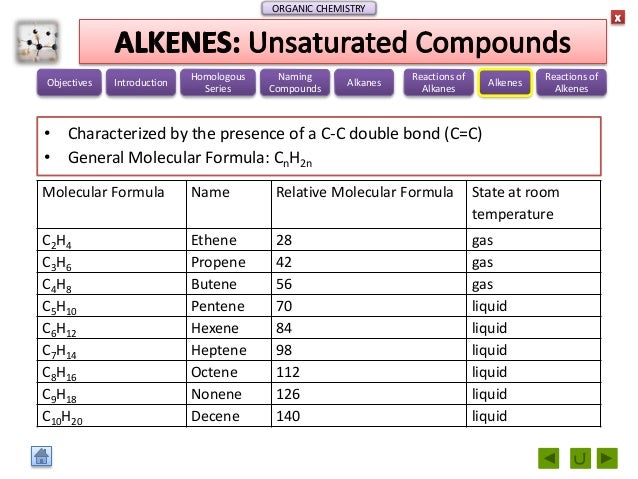
That's why I have drawn this random one above.īut these fragments aren't viable as they stand. They can rotate freely about all the carbon-carbon single bonds, and can take up all sorts of different shapes. We always draw these hydrocarbons as if they were tidy straight chains, but they aren't.  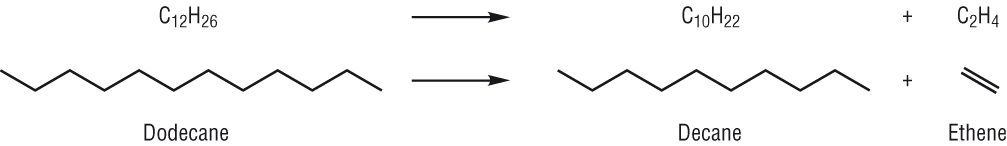
For example, consider the molecule nonane, C 9H 20. The hydrocarbons in crude oil are mainly alkanes and, apart from burning, these aren't actually very reactive.Ĭracking produces molecules containing a carbon-carbon double bond (alkenes) and these are much more reactive and can be turned into useful products.īig molecules are broken at a carbon-carbon single bond. In particular, you need a lot more shorter hydrocarbons in order to meet the demand for petrol (gasoline) than there are in crude oil. The proportions of the various fractions that you get aren't in the same proportion that you ideally need to meet demand. There are two problems with the output from fractional distillation: It looks at a simple experiment to demonstrate cracking in the lab as well as some industrial details.Īlkanes are hydrocarbons which only contain carbon-carbon single bonds.Īlkenes are hydrocarbons which contain a carbon-carbon double bond. This page is about how you can break longer hydrocarbons into more useful shorter ones. Cracking long hydrocarbons to make smaller useful ones
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
